Influence of Carbon Dioxide on Brain Activity
The influence of carbon dioxide on brain activity and metabolism in conscious humans
Feng Xu (1), Jinsoo Uh (1), Matthew R Brier (2), John Hart Jr (2), Uma S Yezhuvath (1), Hong Gu (3), Yihong Yang (3) and Hanzhang Lu (1)
1. Advanced Imaging Research Center, University of Texas Southwestern Medical Center, Dallas, Texas, USA;
2. Center for BrainHealth, University of Texas at Dallas, Dallas, Texas, USA;
3. Neuroimaging Research Branch,National Institute on Drug Abuse, National Institutes of Health, Baltimore, Maryland, USA
A better understanding of carbon dioxide (CO2) effect on brain activity may have a profound impact on clinical studies using CO2 manipulation to assess cerebrovascular reserve and on the use of hypercapnia as a means to calibrate functional magnetic resonance imaging (fMRI) signal.
This study investigates how an increase in blood CO2, via inhalation of 5% CO2, may alter brain activity in humans. Dynamic measurement of brain metabolism revealed that mild hypercapnia resulted in a suppression of cerebral metabolic rate of oxygen (CMRO2) by 13.4%±2.3% (N=14) and, furthermore, the CMRO2 change was proportional to the subject’s end-tidal CO2 (Et-CO2) change. When using functional connectivity MRI (fcMRI) to assess the changes in resting-state neural activity, it was found that hypercapnia resulted in a reduction in all fcMRI indices assessed including cluster volume, cross-correlation coefficient, and amplitude of the fcMRI signal in the default-mode network (DMN).
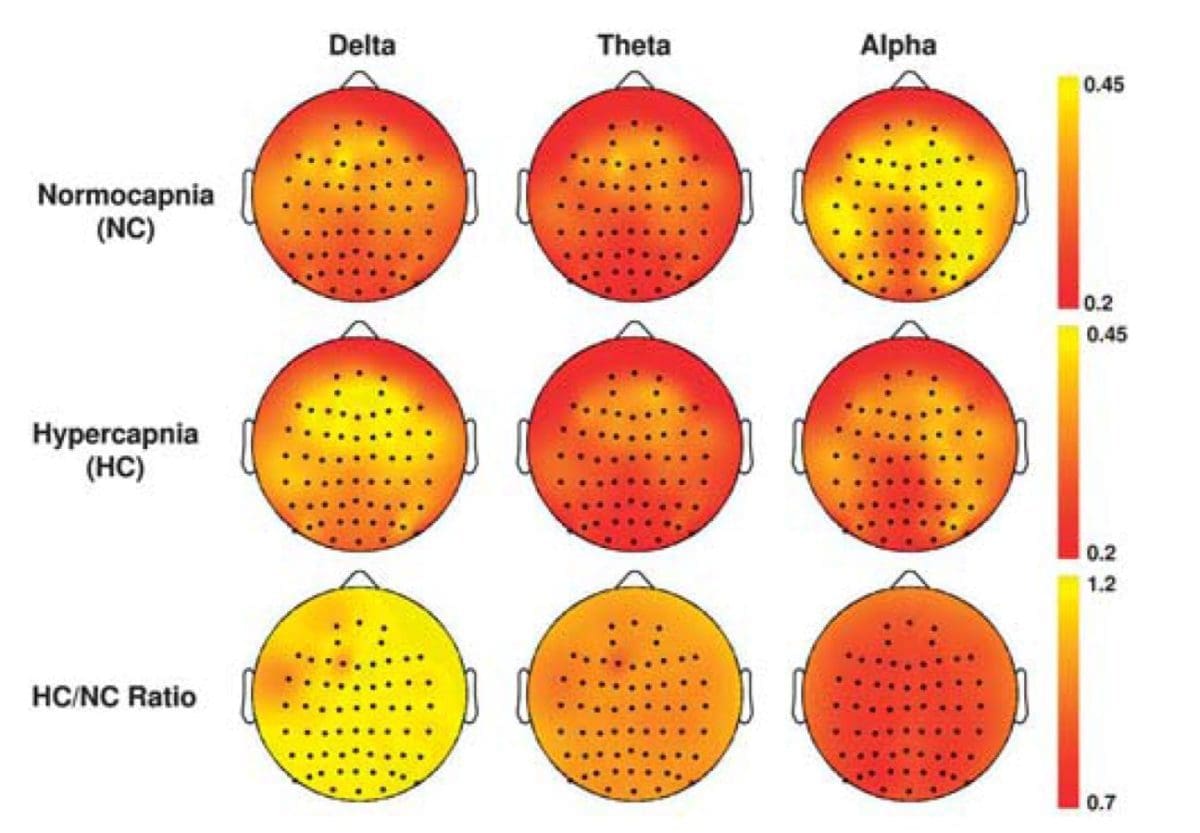
The extent of the reduction was more pronounced than similar indices obtained in visual-evoked fMRI, suggesting a selective suppression effect on resting-state neural activity. Scalp electroencephalogram (EEG) studies comparing hypercapnia with normocapnia conditions showed a relative increase in low frequency power in the EEG spectra, suggesting that the brain is entering a low arousal state on CO2 inhalation.
Discussion
Second, neural activity was assessed indirectly by BOLD fcMRI and the data suggested that CO2 inhalation caused a decrease in spontaneous brain connectivity.
Finally, EEG was used as a direct measure of neural activity and the results showed that hypercapnia caused a relative increase in lower frequency power spectra. Overall, our data showed a suppressive effect of CO2 on brain activity.
Our study represents the first systematic investigation of this effect in conscious humans and suggests that resting-state neural activity is reduced due to mild hypercapnia.
Further, we showed that this suppression of neural activity is accompanied by a reduction in metabolic activity of similar amplitude.
Our observation of a reduced functional connectivity during CO2 breathing is consistent with previous reports that light sedation (Greicius et al, 2008) or anesthesia (Deshpande et al, 2010) decreases connectivity in DMN regions, and is also in line with the suggestions that CO2 has a mild sedative effect (Fukuda et al, 2006).